what will cause the size of the space in the top of the small tube to change in volume
The Beliefs of Gases
Compressibility
- Ascertain compressibility.
- Give examples of the uses of compressed gases.

Will it all fit?
When nosotros pack to go on vacation, in that location is always "i more" thing that we need to get in the suitcase. Mayhap it's another bathing suit, pair of shoes, volume – whatever the item, we need to get it in. Fortunately, we tin can squeeze things together somewhat. There is a piffling space between the folds of clothing, we can rearrange the shoes, and somehow we go that last thing in and close the suitcase.
Compressibility
Scuba diving is a class of underwater diving in which a diver carries his ain breathing gas, normally in the course of a tank of compressed air. The pressure in most commonly used scuba tanks ranges from 200 to 300 atmospheres. Gases are unlike other states of matter in that a gas expands to fill up the shape and book of its container. For this reason, gases can also exist compressed and so that a relatively large corporeality of gas can be forced into a small container. If the air in a typical scuba tank were transferred to a container at the standard pressure of 1 atm, the volume of that container would demand to be near 2500 liters.

Figure xiv.1
Scuba diver.
Compressibility is the measure of how much a given book of affair decreases when placed under force per unit area. If nosotros put force per unit area on a solid or a liquid, at that place is essentially no change in book. The atoms, ions, or molecules that make up the solid or liquid are very close together. At that place is no space betwixt the individual particles, so they cannot pack together.
The kinetic-molecular theory explains why gases are more compressible than either liquids or solids. Gases are compressible because near of the book of a gas is composed of the large amounts of empty space between the gas particles. At room temperature and standard pressure, the average distance between gas molecules is about ten times the diameter of the molecules themselves. When a gas is compressed, as when the scuba tank is being filled, the gas particles are forced closer together.
Compressed gases are used in many situations. In hospitals, oxygen is oft used for patients who have damaged lungs to help them breathe better. If a patient is having a major operation, the anesthesia that is administered will frequently be a compressed gas. Welding requires very hot flames produced by compresses acetylene and oxygen mixtures. Many summer barbeque grills are fueled by compressed propane.

Figure xiv.2
Oxygen tank.
Summary
- Gases volition compress more than easily that solids or liquids because hither is so much space between the gas molecules.
Practice
Questions
Utilise the link below to answer the following questions:
http://world wide web.cdxetextbook.com/engines/motivePower/4gasEng/engcycle.html
- What brings the fuel-air mixture into the cylinder?
- What is the office of the compression cycle?
- Does the frazzle cycle compress the gases produced by ignition?
Review
Questions
- Why is there no modify in book when pressure level is applied to liquids and solids?
- Why do gases compress more hands than liquids and solids?
- List uses for compressed gases.
- compressibility: The measure of how much a given book of affair decreases when placed nether pressure.
Factors Affecting Gas Pressure
- List factors that affect gas force per unit area.
- Explain these effects in terms of the kinetic-molecular theory of gases.

How high does a basketball bounce?
The pressure of the air in a basketball has to exist adjusted and then that the ball bounces to the correct top. Earlier a game, the officials cheque the brawl by dropping information technology from shoulder height and seeing how far dorsum upwards it bounces. What would the official do if the ball did not bounce up as far every bit it is supposed to? What would he practice if it bounced too high?
The pressure level inside a container is dependent on the amount of gas inside the container. If the basketball does not bounce loftier plenty, the official could remedy the situation by using a hand pump and adding more air to the ball. Conversely, if it bounces too high, he could let some air out of the brawl.
Factors Affecting Gas Force per unit area
Call back from the kinetic-molecular theory that gas particles motility randomly and in straight lines until they elastically collide with either other gas particles or with one of the walls of the container. It is these collisions with the walls of the container that defines the pressure level of the gas. Four variables are used to describe the condition of a gas. They are pressure level ![]() , book
, book ![]() , temperature
, temperature ![]() , and the amount of the gas equally measured by the number moles
, and the amount of the gas equally measured by the number moles ![]() . Nosotros will examine separately how the volume, temperature, and amount of gas each affect the pressure of an enclosed gas sample.
. Nosotros will examine separately how the volume, temperature, and amount of gas each affect the pressure of an enclosed gas sample.
Corporeality of Gas
The Effigy beneath shows what happens when air is added to a rigid container . A rigid container is one that is incapable of expanding or contracting. A steel canister is an instance of a rigid container.

Figure xiv.iii
Increment in force per unit area with increase in number of gas particles.
The canister on the left contains a gas at a certain pressure. The attached air pump is then used to double the amount of gas in the canister. Since the canister cannot aggrandize, the increased number of air molecules volition strike the inside walls of the canister twice as oftentimes as they did before. The result is that the pressure inside the canister doubles. As you might imagine, if more and more air is continually added to a rigid container, it may eventually burst. Reducing the number of molecules in a rigid container has the opposite outcome and the pressure decreases.
Volume
Pressure is besides afflicted by the volume of the container. If the volume of a container is decreased, the gas molecules have less space in which to movement around. As a result, they volition strike the walls of the container more oft and the pressure increases.
Figure below shows a cylinder of gas whose volume is controlled by an adjustable piston. On the left, the piston is pulled mostly out and the gauge reads a certain pressure. On the right, the piston has been pushed so that the volume of the enclosed portion of the container where the gas is located has been cut in half. The pressure level of the gas doubles. Increasing the book of the container would take the contrary issue and the pressure level of the gas would decrease.

Effigy 14.four
Decrease in gas volume produced increase in gas pressure.
Temperature
It would be very unadvisable to place a can of soup over a campfire without venting the can. Equally the can heats up, it may explode. The kinetic-molecular theory explains why. The air within the rigid tin can of soup is given more kinetic energy past the heat coming from the bivouac. The kinetic energy causes the air molecules to move faster and they impact the container walls more frequently and with more force. The increment in pressure level within may eventually exceed the forcefulness of the can and it will explode. An boosted factor is that the soup may begin boiling which will then aid fifty-fifty more gas and more pressure to the inside of the tin can.
Shown in the Figure below is a cylinder of gas on the left that is at room temperature (300 K). On the right, the cylinder has been heated until the Kelvin temperature has doubled to 600 One thousand. The kinetic energy of the gas molecules increases, so collisions with the walls of the container are now more than forceful than they were earlier. As a result, the pressure of the gas doubles. Decreasing the temperature would have the reverse consequence, and the pressure level of an enclosed gas would decrease.

Figure fourteen.v
Increase in temperature produces increase in pressure.
Summary
- An increment in the number of gas molecules in the aforementioned volume container increases force per unit area.
- A decrease in container book increases gas pressure.
- An increase in temperature of a gas in a rigid container increases the pressure level.
Practise
Questions
Lookout the video at the link below and respond the post-obit questions:

Click on the image above for more content
http://www.youtube.com/watch?5=0mVuWZ7nvcU
- What causes pressure?
- What happens when you allow gas out of the container?
- If you increase the temperature, what happens to the pressure?
- Why does the pressure driblet when you increase the book?
Review
Questions
- What defines the pressure of a gas?
- Why does an increase in the number of molecules increase the force per unit area?
- Why does an increment in temperature increase the pressure?
- rigid container: One that is incapable of expanding or contracting.
Boyle's Law
- State Boyle'due south Constabulary.
- Use Boyle'south Police force to summate volume-pressure level relationships.

How important is it to check the weather?
Each mean solar day, hundreds of weather balloons are launched. Made of a synthetic safe and conveying a box of instruments, the helium-filled balloon rises up into the heaven. Equally it gains altitude, the atmospheric force per unit area becomes less and the balloon expands. At some point the balloon bursts due to the expansion, the instruments driblet (aided by a parachute) to exist retrieved and studied for information about the weather.
Boyle's Law
Robert Boyle (1627-1691), an English language chemist, is widely considered to be one of the founders of the modern experimental science of chemistry. He discovered that doubling the pressure of an enclosed sample of gas while keeping its temperature constant acquired the volume of the gas to be reduced by half. Boyle's law states that the volume of a given mass of gas varies inversely with the pressure when the temperature is kept constant. An inverse human relationship is described in this way. As ane variable increases in value, the other variable decreases.
Physically, what is happening? The gas molecules are moving and are a sure altitude autonomously from one some other. An increment in pressure pushes the molecules closer together, reducing the volume. If the pressure is decreased, the gases are free to move about in a larger volume.

Figure 14.6
Robert Boyle.
Mathematically, Boyle's law can be expressed by the equation:
![]()
The ![]() is a abiding for a given sample of gas and depends only on the mass of the gas and the temperature. The Table below shows pressure and volume data for a set amount of gas at a constant temperature. The third column represents the value of the constant
is a abiding for a given sample of gas and depends only on the mass of the gas and the temperature. The Table below shows pressure and volume data for a set amount of gas at a constant temperature. The third column represents the value of the constant ![]() for this data and is always equal to the pressure level multiplied past the volume. As one of the variables changes, the other changes in such a style that the product of
for this data and is always equal to the pressure level multiplied past the volume. As one of the variables changes, the other changes in such a style that the product of ![]() always remains the same. In this particular case, that abiding is 500 atm · ml.
always remains the same. In this particular case, that abiding is 500 atm · ml.
| Pressure (atm) | Volume (mL) | |
| 0.5 | m | 500 |
| 0.625 | 800 | 500 |
| ane.0 | 500 | 500 |
| 2.0 | 250 | 500 |
| 5.0 | 100 | 500 |
| 8.0 | 62.5 | 500 |
| 10.0 | fifty | 500 |
A graph of the data in the table farther illustrates the changed relationship nature of Boyle'due south Law (meet Figure below ). Book is plotted on the ![]() -axis, with the corresponding pressure on the
-axis, with the corresponding pressure on the ![]() -axis.
-axis.

Figure 14.seven
The pressure of a gas decreases as the volume increases, making Boyle's law an inverse relationship.
Boyle'southward Constabulary can exist used to compare irresolute conditions for a gas. Nosotros utilise ![]() and
and ![]() to stand for the initial pressure and initial volume of a gas. After a modify has been made,
to stand for the initial pressure and initial volume of a gas. After a modify has been made, ![]() and
and ![]() correspond the terminal pressure and volume. The mathematical relationship of Boyle's Law becomes:
correspond the terminal pressure and volume. The mathematical relationship of Boyle's Law becomes:
![]()
This equation tin be used to calculate whatsoever one of the four quantities if the other 3 are known.
Sample Problem: Boyle'southward Police
A sample of oxygen gas has a volume of 425 mL when the force per unit area is equal to 387 kPa. The gas is allowed to expand into a 1.75 L container. Summate the new pressure of the gas.
Step one: List the known quantities and program the problem.
Known
Unknown
Use Boyle's Police force to solve for the unknown force per unit area ![]() . It is of import that the two volumes (
. It is of import that the two volumes ( ![]() and
and ![]() ) are expressed in the same units, and then
) are expressed in the same units, and then ![]() has been converted to mL.
has been converted to mL.
Step 2: Solve.
Kickoff, rearrange the equation algebraically to solve for ![]() .
.
![]()
Now substitute the known quantities into the equation and solve.
![]()
Step 3: Think nigh your result.
The volume has increased to slightly over four times its original value and so the pressure is decreased past about ![]() . The pressure level is in kPa and the value has three pregnant figures. Note that any pressure level or book units can exist used as long as they are consistent throughout the problem.
. The pressure level is in kPa and the value has three pregnant figures. Note that any pressure level or book units can exist used as long as they are consistent throughout the problem.
Summary
- The volume of a gas is inversely proportional to temperature.
Practice
Do the problems at the link beneath:
http://www.concord.org/~ddamelin/chemsite/g_gasses/handouts/Boyle_Problems.pdf
Review
Questions
- What does "inversely" hateful in this law?
- Explicate Boyle's law in terms of the kinetic-molecular theory of gases.
- Does it thing what units are used?
- Boyle'southward law: The volume of a given mass of gas varies inversely with the pressure when the temperature is kept constant.
Charles's Police force
- State Charles' Police.
- Use this law to perform calculations involving volume-temperature relationships.

How do you broil breadstuff?
Everybody enjoys the smell and taste of freshly-baked bread. It is low-cal and fluffy as a consequence of the action of yeast on sugar. The yeast converts the sugar to carbon dioxide, which at high temperatures causes the dough to expand. The end-upshot is an enjoyable treat, especially when covered with melted butter.
Charles's Law

Figure 14.eight
Equally a container of confined gas is heated, its molecules increment in kinetic energy and push the movable piston outward, resulting in an increase in book.
French physicist Jacques Charles (1746-1823) studied the event of temperature on the book of a gas at abiding pressure. Charles's law states that the volume of a given mass of gas varies direct with the accented temperature of the gas when pressure is kept abiding. The accented temperature is temperature measured with the Kelvin scale. The Kelvin calibration must be used because zero on the Kelvin calibration corresponds to a complete stoppage of molecular motility.
Mathematically, the direct relationship of Charles's law tin can be represented past the following equation:
![]()
As with Boyle'southward law, ![]() is constant only for a given gas sample. The Table below shows temperature and volume data for a set up amount of gas at a constant pressure level. The third cavalcade is the constant for this particular data set and is e'er equal to the volume divided by the Kelvin temperature.
is constant only for a given gas sample. The Table below shows temperature and volume data for a set up amount of gas at a constant pressure level. The third cavalcade is the constant for this particular data set and is e'er equal to the volume divided by the Kelvin temperature.
| Temperature (K) | Volume (mL) | |
| 50 | 20 | 0.40 |
| 100 | 40 | 0.twoscore |
| 150 | 60 | 0.40 |
| 200 | lxxx | 0.40 |
| 300 | 120 | 0.40 |
| 500 | 200 | 0.forty |
| 1000 | 400 | 0.xl |
When this data is graphed, the result is a straight line, indicative of a straight human relationship, shown in Effigy below .

Effigy 14.ix
The book of a gas increases every bit the Kelvin temperature increases.
Notice that the line goes exactly toward the origin, meaning that equally the absolute temperature of the gas approaches zero, its book approaches cypher. Nonetheless, when a gas is brought to extremely common cold temperatures, its molecules would somewhen condense into the liquid state earlier reaching accented zero. The temperature at which this change into the liquid state occurs varies for dissimilar gases.
Charles'southward Police force can also be used to compare changing conditions for a gas. Now we use ![]() and
and ![]() to stand for the initial volume and temperature of a gas, while
to stand for the initial volume and temperature of a gas, while ![]() and
and ![]() represent the final volume and temperature. The mathematical relationship of Charles'southward Constabulary becomes:
represent the final volume and temperature. The mathematical relationship of Charles'southward Constabulary becomes:
![]()
This equation can be used to calculate whatever i of the 4 quantities if the other iii are known. The direct human relationship will just concur if the temperatures are expressed in Kelvin. Temperatures in Celsius volition non work. Remember the human relationship that K = °C + 273.
Sample Trouble: Charles'south Law
A balloon is filled to a volume of two.20 Fifty at a temperature of 22°C. The balloon is so heated to a temperature of 71°C. Find the new volume of the balloon.
Step 1: List the known quantities and plan the problem.
Known
Unknown
Use Charles'south police to solve for the unknown volume ![]() . The temperatures accept first been converted to Kelvin.
. The temperatures accept first been converted to Kelvin.
Step 2: Solve.
Kickoff, rearrange the equation algebraically to solve for ![]() .
.
![]()
Now substitute the known quantities into the equation and solve.
![]()
Step iii: Think nigh your result.
The volume increases as the temperature increases. The consequence has three significant figures.
Summary
- Increasing the temperature of a gas at constant pressure level will produce and increase in the volume.
Practice
Perform the calculations at the web site below:
http://mmsphyschem.com/chuckL.pdf
Review
Questions
- Explain Charles' Law in terms of the kinetic molecular theory.
- Why does the temperature need to exist in Kelvin?
- Does Charles' law hold when the gas becomes a liquid?
- Charles's law: The volume of a given mass of gas varies directly with the absolute temperature of the gas when pressure is kept constant.
Gay-Lussac's Law
- State Gay-Lussac's police.
- Use this police force to perform calculations involving force per unit area-temperature relationships.

How much propane is in the tank?
Propane tanks are widely used with barbeque grills. But it'southward not fun to find out half-way through your grilling that y'all've run out of gas. You can buy gauges that measure the force per unit area inside the tank to see how much is left. The gauge measures pressure and will annals a higher pressure on a hot twenty-four hours than it will on a cold day. So you need to take the air temperature into account when you determine whether or not to refill the tank before your next cook-out.
Gay-Lussac'south Law
When the temperature of a sample of gas in a rigid container is increased, the force per unit area of the gas increases also. The increase in kinetic energy results in the molecules of gas striking the walls of the container with more force, resulting in a greater pressure. The French chemist Joseph Gay-Lussac (1778-1850) discovered the human relationship between the pressure of a gas and its absolute temperature. Gay-Lussac's constabulary states that the pressure of a given mass of gas varies directly with the absolute temperature of the gas, when the volume is kept constant. Gay-Lussac's law is very like to Charles's police, with the merely difference being the type of container. Whereas the container in a Charles'due south law experiment is flexible, it is rigid in a Gay-Lussac's law experiment.

Figure 14.10
Joseph Louis Gay-Lussac.
The mathematical expressions for Gay-Lussac'south police force are as well similar to those of Charles's law:
![]()
A graph of pressure vs. temperature also illustrates a directly relationship. As a gas is cooled at constant volume its pressure continually decreases until the gas condenses to a liquid.
Sample Problem: Gay-Lussac'south Law
The gas in an aerosol can is nether a pressure of three.00 atm at a temperature of 25°C. It is unsafe to dispose of an aerosol tin by incineration. What would the pressure in the aerosol tin can be at a temperature of 845°C?
Step 1: List the known quantities and plan the trouble.
Known
Unknown
Use Gay-Lussac's law to solve for the unknown pressure ![]() . The temperatures have first been converted to Kelvin.
. The temperatures have first been converted to Kelvin.
Step 2: Solve.
First, rearrange the equation algebraically to solve for ![]() .
.
![]()
Now substitute the known quantities into the equation and solve.
![]()
Stride 3: Think well-nigh your result.
The pressure increases dramatically due to large increase in temperature.
Summary
- Pressure and temperature at constant volume are direct proportional.
Practice
Piece of work on the problems establish at the web site beneath:
http://world wide web.chemteam.info/GasLaw/WS-Gay-Lussac.html
Review
Questions
- Explain Gay-Lussac's Law in terms of the kinetic-molecular theory.
- What would a graph of pressure vs. temperature testify us?
- What is the difference in containers in Charles' Constabulary and Gay-Lussac's Law?
- Gay-Lussac's police: The pressure of a given mass of gas varies directly with the accented temperature of the gas, when the volume is kept constant.
Combined Gas Law
- Country the combined gas police force.
- Use the police force to calculate parameters in full general gas problems.

What keeps things cold?
The modern fridge takes advantage of the gas laws to remove heat from a organisation. Compressed gas in the coils (see higher up) is immune to expand. This expansion lowers the temperature of the gas and transfers heat energy from the material in the refrigerator to the gas. Equally the gas is pumped through the coils, the pressure on the gas compresses it and raises the gas temperature. This heat is then dissipated through the coils into the exterior air. Every bit the compressed gas is pumped through the system over again, the procedure repeats itself.
Combined Gas Constabulary
To this indicate, we have examined the relationships between whatsoever two of the variables of ![]() ,
, ![]() , and
, and ![]() , while the third variable is held constant. However, situations arise where all iii variables alter. The combined gas law expresses the human relationship between the pressure, volume, and absolute temperature of a stock-still amount of gas. For a combined gas law problem, only the amount of gas is held constant.
, while the third variable is held constant. However, situations arise where all iii variables alter. The combined gas law expresses the human relationship between the pressure, volume, and absolute temperature of a stock-still amount of gas. For a combined gas law problem, only the amount of gas is held constant.
![]()
Sample Trouble: Combined Gas Police
2.00 Fifty of a gas at 35°C and 0.833 atm is brought to standard temperature and pressure (STP). What will be the new gas volume?
Step ane: List the known quantities and plan the trouble.
Known
Unknown
Use the combined gas law to solve for the unknown volume ![]() . STP is 273 K and i atm. The temperatures have been converted to Kelvin.
. STP is 273 K and i atm. The temperatures have been converted to Kelvin.
Step 2: Solve
First, rearrange the equation algebraically to solve for ![]() .
.
![]()
Now substitute the known quantities into the equation and solve.
![]()
Pace 3: Think about your result.
Both the increment in pressure and the decrease in temperature cause the volume of the gas sample to decrease. Since both changes are relatively minor, the volume does non subtract dramatically.
It may seem challenging to recall all the different gas laws introduced and so far. Fortunately, Boyle'southward, Charles'due south, and Gay-Lussac'south laws can all be easily derived from the combined gas police force. For instance, consider a state of affairs where a change occurs in the volume and pressure of a gas while the temperature is beingness held constant. In that case, it can be said that ![]() . Look at the combined gas law and cancel the
. Look at the combined gas law and cancel the ![]() variable out from both sides of the equation. What is left over is Boyle's law:
variable out from both sides of the equation. What is left over is Boyle's law:
![]() . Too, if the pressure is constant, and so
. Too, if the pressure is constant, and so ![]() and canceling
and canceling ![]() out of the equation leaves Charles's police. If the book is constant, then
out of the equation leaves Charles's police. If the book is constant, then ![]() and canceling
and canceling ![]() out of the equation leaves Gay-Lussac's constabulary.
out of the equation leaves Gay-Lussac's constabulary.
Summary
- The combined gas police shows the relationships amongst temperature, volume, and pressure.
-

Practice
Piece of work on the problems at the link below:
http://misterguch.brinkster.cyberspace/WKS001_007_146637.pdf
Review
Questions
- What is the only thing held constant in a combined gas constabulary problem?
- If you want to solve for the volume of a gas
 and
and  is greater than
is greater than  , would you expect
, would you expect  to be larger or smaller than
to be larger or smaller than  ?
? - What would exist the equation for finding
 given all the other parameters?
given all the other parameters?
- combined gas law: Expresses the relationship between the pressure, book, and absolute temperature of a fixed amount of gas.
Avogadro'southward Police force
- State Avogadro'south Law.
- Apply this law to perform calculations involving quantities of gases.

How much air exercise you put into a tire?
A flat tire is not very useful. It does non cushion the rim of the wheel and creates a very uncomfortable ride. When air is added to the tire, the pressure increases as more molecules of gas are forced into the rigid tire. How much air should be put into a tire depends on the pressure rating for that tire. Too little pressure level and the tire will non concur its shape. Too much pressure and the tire could flare-up.
Avogadro'due south Law
You take learned well-nigh Avogadro'south hypothesis: equal volumes of whatever gas at the same temperature and pressure comprise the aforementioned number of molecules. It follows that the volume of a gas is direct proportional to the number of moles of gas nowadays in the sample. Avogadro's law states that the volume of a gas is directly proportional to the number of moles of gas when the temperature and pressure are held abiding. The mathematical expression of Avogadro's constabulary is
![]()
where ![]() is the number of moles of gas and
is the number of moles of gas and ![]() is a constant. Avogadro's police is in evidence whenever you blow up a airship. The book of the airship increases as you lot add moles of gas to the balloon by blowing it upwardly.
is a constant. Avogadro's police is in evidence whenever you blow up a airship. The book of the airship increases as you lot add moles of gas to the balloon by blowing it upwardly.
If the container holding the gas is rigid rather than flexible, pressure can be substituted for volume in Avogadro'due south police force. Adding gas to a rigid container makes the pressure increase.
Sample Problem: Avogadro's Constabulary
A balloon has been filled to a volume of 1.ninety L with 0.0920 mol of helium gas. If 0.0210 mol of additional helium is added to the balloon while the temperature and pressure level are held constant, what is the new volume of the balloon?
Footstep one: List the known quantities and plan the trouble.
Known
Unknown
Annotation that the terminal number of moles has to be calculated by adding the original number of moles to the moles of added helium. Employ Avogadro'due south law to solve for the final volume.
Footstep 2: Solve.
First, rearrange the equation algebraically to solve for ![]() .
.
![]()
Now substitute the known quantities into the equation and solve.
![]()
Stride 3: Think almost your effect.
Since a relatively minor corporeality of additional helium was added to the balloon, its volume increases slightly.
Summary
- Calculations are shown for relationships between volume and number of moles of a gas.
Practice
Piece of work on the problems at the site below:
http://www.gst-d2l.com/homework/hwavogadroslaw.html
Review
Questions
- What is held constant in the Avogadro'southward Law human relationship?
- What happens if you add gas to a rigid container?
- Why does a balloon expand when you add air to information technology?
- Avogadro's law: The volume of a gas is directly proportional to the number of moles of gas when the temperature and pressure are held abiding.
Platonic Gas Law
- Derive the ideal gas law from the combined gas constabulary and Avogadro's law.
- Calculate the value of the ideal gas constant.
- Use the ideal gas police to calculate parameters for ideal gases.

What chemical reactions require ammonia?
There are a number of chemical reactions that crave ammonia. In order to carry out the reaction efficiently, we demand to know how much ammonia we have for stoichiometric purposes. Using gas laws, we tin can decide the number of moles present in the tank if we know the volume, temperature, and force per unit area of the organization.
Platonic Gas Police force
The combined gas law shows that the pressure of a gas is inversely proportional to book and directly proportional to temperature. Avogadro's law shows that volume or force per unit area is direct proportional to the number of moles of gas. Putting these together leaves us with the post-obit equation:
![]()
As with the other gas laws, we can also say that ![]() is equal to a constant. The constant can be evaluated provided that the gas being described is considered to be ideal.
is equal to a constant. The constant can be evaluated provided that the gas being described is considered to be ideal.
The ideal gas law is a single equation which relates the pressure, volume, temperature, and number of moles of an ideal gas. If we substitute in the variable ![]() for the constant, the equation becomes:
for the constant, the equation becomes:
![]()
The ideal gas law is conventionally rearranged to look this way, with the multiplication signs omitted:
![]()
The variable ![]() in the equation is called the platonic gas abiding .
in the equation is called the platonic gas abiding .
Evaluating the Ideal Gas Constant
The value of ![]() , the ideal gas abiding, depends on the units chosen for pressure, temperature, and book in the platonic gas equation. Information technology is necessary to use Kelvin for the temperature and it is conventional to employ the SI unit of measurement of liters for the book. However, force per unit area is ordinarily measured in i of 3 units: kPa, atm, or mmHg. Therefore,
, the ideal gas abiding, depends on the units chosen for pressure, temperature, and book in the platonic gas equation. Information technology is necessary to use Kelvin for the temperature and it is conventional to employ the SI unit of measurement of liters for the book. However, force per unit area is ordinarily measured in i of 3 units: kPa, atm, or mmHg. Therefore, ![]() can have three different values.
can have three different values.
We will demonstrate how ![]() is calculated when the pressure level is measured in kPa. Call up that the volume of 1.00 mol of any gas at STP is measured to be 22.414 L. We tin substitute 101.325 kPa for pressure level, 22.414 L for volume, and 273.xv Chiliad for temperature into the ideal gas equation and solve for
is calculated when the pressure level is measured in kPa. Call up that the volume of 1.00 mol of any gas at STP is measured to be 22.414 L. We tin substitute 101.325 kPa for pressure level, 22.414 L for volume, and 273.xv Chiliad for temperature into the ideal gas equation and solve for ![]() .
.
![]()
This is the value of ![]() that is to be used in the ideal gas equation when the pressure level is given in kPa. The Table below shows a summary of this and the other possible values of
that is to be used in the ideal gas equation when the pressure level is given in kPa. The Table below shows a summary of this and the other possible values of ![]() . It is important to choose the right value of
. It is important to choose the right value of ![]() to use for a given trouble.
to use for a given trouble.
| Unit of | Unit of | Unit of | Unit of | Value and unit of measurement of |
| kPa | Fifty | mol | K | |
| atm | Fifty | mol | One thousand | |
| mmHg | 50 | mol | K | |
Notice that the unit of measurement for ![]() when the pressure is in kPa has been changed to J/K • mol. A kilopascal multiplied by a liter is equal to the SI unit for energy, a joule (J).
when the pressure is in kPa has been changed to J/K • mol. A kilopascal multiplied by a liter is equal to the SI unit for energy, a joule (J).
Sample Problem: Ideal Gas Law
What book is occupied past 3.760 yard of oxygen gas at a pressure level of 88.four kPa and a temperature of xix°C? Presume the oxygen is ideal.
Step 1: List the known quantities and programme the problem.
Known
Unknown
In society to employ the ideal gas police, the number of moles of O two ![]() must exist plant from the given mass and the molar mass. Then, utilize
must exist plant from the given mass and the molar mass. Then, utilize ![]() to solve for the volume of oxygen.
to solve for the volume of oxygen.
Step two: Solve .
![]()
Rearrange the platonic gas police and solve for ![]() .
.
![]()
Pace iii: Think virtually your result
The number of moles of oxygen is far less than one mole, then the volume should exist fairly small compared to molar volume (22.iv L/mol) since the force per unit area and temperature are reasonably close to standard. The issue has three significant figures because of the values for ![]() and
and ![]() . Since a joule (J) = kPa • L, the units cancel correctly, leaving a volume in liters.
. Since a joule (J) = kPa • L, the units cancel correctly, leaving a volume in liters.
Summary
- The ideal gas constant is calculated.
- An example of calculations using the platonic gas law is shown.
Exercise
Piece of work on the problems at the link below:
http://chemsite.lsrhs.net/gasses/handouts/Ideal_Problems.pdf
Review
Questions
- Which value of
 will you use if the pressure level is given in atm?
will you use if the pressure level is given in atm? - Yous are doing a calculation where the pressure is given in mm Hg. You lot select viii.314 J/Yard • mol equally your value for
 . Will yous get a correct answer?
. Will yous get a correct answer? - How would y'all cheque that you accept chosen the correct value of
 for your trouble?
for your trouble?
- ideal gas abiding: The variable
 in the ideal gas police force equation.
in the ideal gas police force equation. - ideal gas police force: A single equation which relates the pressure level, volume, temperature, and number of moles of an platonic gas.
Calculating the Molar Mass of a Gas
- Calculate the molar mass of a gas.
- Summate the density of a gas.

What makes it float?
Helium has long been used in balloons and blimps. Since it is much less dense than air, it will bladder higher up the ground. We tin can buy small balloons filled with helium at stores, but large ones (such as the balloon seen higher up) are much more expensive and take up a lot more helium.
Calculating Molar Mass and Density of a Gas
A chemical reaction, which produces a gas, is performed. The produced gas is and then collected and its mass and volume are determined. The molar mass of the unknown gas tin be establish using the ideal gas law, provided the temperature and pressure of the gas are besides known.
Sample Problem: Molar Mass and the Platonic Gas Law
A sure reaction occurs, producing an oxide of nitrogen as a gas. The gas has a mass of i.211 g and occupies a volume of 677 mL. The temperature in the laboratory is 23°C and the air pressure level is 0.987 atm. Summate the molar mass of the gas and deduce its formula. Assume the gas is ideal.
Step 1: List the known quantities and programme the problem .
Known
Unknown
Get-go the ideal gas law will be used to solve for the moles of unknown gas ![]() . And so the mass of the gas divided by the moles volition give the tooth mass.
. And so the mass of the gas divided by the moles volition give the tooth mass.
Stride two: Solve .
![]()
At present separate chiliad by mol to get the molar mass.
![]()
Since Northward has a molar mass of 14 yard/mol and O has a tooth mass of xvi g/mol, the formula N ii O would produce the correct molar mass.
Step 3: Retrieve about your result
The ![]() value that corresponds to a pressure level in atm was chosen for this trouble. The calculated molar mass gives a reasonable formula for dinitrogen monoxide.
value that corresponds to a pressure level in atm was chosen for this trouble. The calculated molar mass gives a reasonable formula for dinitrogen monoxide.
Calculating Density of a Gas
The ideal gas police can be used to find the density of a gas at atmospheric condition that are non standard. For case, we will determine the density of ammonia gas (NH 3 ) at 0.913 atm and 20°C, assuming the ammonia is ideal. First, the molar mass of ammonia is calculated to be 17.04 g/mol. Next, assume exactly 1 mol of ammonia ![]() and calculate the volume that such an amount would occupy at the given temperature and pressure.
and calculate the volume that such an amount would occupy at the given temperature and pressure.
![]()
Now the density tin can be calculated by dividing the mass of one mole of ammonia by the volume above.
![]()
As a point of comparison, this density is slightly less than the density of ammonia at STP, which is equal to ![]() . It makes sense that the density should be lower compared to that at STP since both the increase in temperature (from 0°C to twenty°C) and the subtract in pressure level (from one atm to 0.913 atm) would cause the NH 3 molecules to spread out a bit farther from one another.
. It makes sense that the density should be lower compared to that at STP since both the increase in temperature (from 0°C to twenty°C) and the subtract in pressure level (from one atm to 0.913 atm) would cause the NH 3 molecules to spread out a bit farther from one another.
Summary
- Calculations of molar mass and density of an ideal gas are described.
Practise
Answer questions and perform calculations of problems at the post-obit link:
http://www.mybookezz.com/ebook.php?u=aHR0cDovL2dvLmhydy5jb20vcmVzb3VyY2VzL2dvX3NjL21jL0hDMlNSMTEzLlBERgpTZWN0aW9uIDM=
Review
Questions
- Why do you need the volume, temperature, and pressure of the gas to calculate molar mass?
- What assumption most the gas is made in all these calculations?
- Why exercise y'all need the mass of the gas to summate the tooth mass?
Gas Stoichiometry
- Utilise the platonic gas law to calculate stoichiometry problems for gases.

How is fertilizer produced?
The Haber bicycle reaction of gaseous nitrogen and hydrogen to form ammonia is a critical step in the production of fertilizer from ammonia. It is important to accept an backlog of the starting materials so that a maximum yield of ammonia can be accomplished. Past knowing how much ammonia is needed for manufacture of a batch of fertilizer, the proper amounts of nitrogen and hydrogen gases can be incorporated into the process.
Gas Stoichiometry
You have learned how to employ tooth book to solve stoichiometry problems for chemical reactions involving ane or more than gases at STP. At present, nosotros can utilize the ideal gas law to expand our treatment of chemical reactions to solve stoichiometry bug for reactions that occur at any temperature and pressure.
Sample Problem: Gas Stoichiometry and the Platonic Gas Police force
What volume of carbon dioxide is produced past the combustion of 25.21 g of ethanol (C 2 H five OH) at 54°C and 728 mmHg? Assume the gas is ideal.
Before using the ideal gas law, information technology is necessary to write and residual the chemical equation. Recall that most combustion reactions, the given substance reacts with O two to grade CO ii and H two O. Here is the counterbalanced equation for the combustion of ethanol.
![]()
Step 1: List the known quantities and solve the problem.
Known
Unknown
The number of moles of carbon dioxide gas is showtime calculated by stoichiometry. Then the ideal gas law is used to summate the volume of CO 2 produced.
Footstep two: Solve.
![]()
The moles of ethanol ![]() is now substituted into
is now substituted into ![]() to solve for the volume.
to solve for the volume.
![]()
Step 3: Think about your event.
The mass of ethanol is slightly more than than ane half mole, meaning that the mole ratio results in slightly more than than one mole of carbon dioxide being produced. Because of the elevated temperature and reduced pressure compared to STP, the resulting volume is larger than 22.4 Fifty.
Summary
- The ideal gas law is used to summate stoichiometry issues for gases.
Practice
Solve the problems on the worksheet at this site:
http://misterguch.brinkster.net/PRA036.pdf
Review
Questions
- Do we demand gas conditions to be at STP to calculate stoichiometry problems?
- Why exercise nosotros want to determine the stoichiometry of these reactions?
- What assumption are we making about the gases involved?
Existent and Ideal Gases
- Define a existent gas.
- Depict differences betwixt real gases and platonic gases.

Location, Location, Location
The behavior of a molecule depends a lot on its construction. We tin have two compounds with the aforementioned number of atoms and nevertheless they act very differently. Ethanol (C 2 H v OH) is a clear liquid that has a humid point of about 79°C. Dimethylether (CH iii OCH 3 ) has the aforementioned number of carbons, hydrogens, and oxygens, only boils at a much lower temperature (-25°C). The difference lies in the amount of intermolecular interaction (strong H-bonds for ethanol, weak van der Waals force for the ether).
Real and Platonic Gases
An ideal gas is one that follows the gas laws at all conditions of temperature and pressure. To do so, the gas would need to completely abide past the kinetic-molecular theory. The gas particles would demand to occupy zero volume and they would need to exhibit no bonny forces what then ever toward each other. Since neither of those conditions can be true, there is no such thing equally an platonic gas. A existent gas is a gas that does not behave according to the assumptions of the kinetic-molecular theory. Fortunately, at the weather condition of temperature and force per unit area that are normally encountered in a laboratory, existent gases tend to behave very much like ideal gases.
Under what conditions then, do gases comport least ideally? When a gas is put nether loftier pressure, its molecules are forced closer together every bit the empty space between the particles is macerated. A decrease in the empty space ways that the assumption that the book of the particles themselves is negligible is less valid. When a gas is cooled, the decrease in kinetic energy of the particles causes them to wearisome down. If the particles are moving at slower speeds, the attractive forces between them are more prominent. Another mode to view it is that connected cooling the gas volition eventually plow it into a liquid and a liquid is certainly not an ideal gas anymore (see liquid nitrogen in the Figure beneath ). In summary, a real gas deviates virtually from an platonic gas at low temperatures and high pressures. Gases are well-nigh ideal at high temperature and depression pressure level.

Figure fourteen.11
Nitrogen gas that has been cooled to 77 K has turned to a liquid and must be stored in a vacuum insulated container to prevent it from chop-chop vaporizing.
The Effigy below shows a graph of ![]() plotted confronting pressure for 1 mol of a gas at iii different temperatures – 200 G, 500 K, and yard K. An ideal gas would take a value of ane for that ratio at all temperatures and pressures and the graph would simply be a horizontal line. As can exist seen, deviations from an platonic gas occur. Equally the pressure level begins to ascension, the attractive forces cause the book of the gas to be less than expected and the value of
plotted confronting pressure for 1 mol of a gas at iii different temperatures – 200 G, 500 K, and yard K. An ideal gas would take a value of ane for that ratio at all temperatures and pressures and the graph would simply be a horizontal line. As can exist seen, deviations from an platonic gas occur. Equally the pressure level begins to ascension, the attractive forces cause the book of the gas to be less than expected and the value of ![]() drops nether i. Continued pressure increase results in the volume of the particles to become significant and the value of
drops nether i. Continued pressure increase results in the volume of the particles to become significant and the value of ![]() rises to greater than 1. Observe, that the magnitude of the deviations from ideality is greatest for the gas at 200 Thou and least for the gas at 1000 Thou.
rises to greater than 1. Observe, that the magnitude of the deviations from ideality is greatest for the gas at 200 Thou and least for the gas at 1000 Thou.

Figure 14.12
Real gases deviate from ideal gases at high pressures and at low temperatures.
The ideality of a gas also depends on the strength and type of intermolecular attractive forces that be between the particles. Gases whose bonny forces are weak are more than ideal than those with stiff attractive forces. At the same temperature and pressure, neon is more platonic than water vapor considering neon's atoms are but attracted by weak dispersion forces, while water vapor's molecules are attracted by relatively stronger hydrogen bonds. Helium is a more than ideal gas than neon considering its smaller number of electrons means that helium's dispersion forces are fifty-fifty weaker than those of neon.
Summary
- The backdrop of existent gases and their deviations from ideality are described.
Practice
Questions
Employ the link beneath to answer the following questions:
http://world wide web.adichemistry.com/physical/gaseous/deviation/van-der-waals-equation.html
- What is the compressibility factor for a perfect (platonic) gas?
- What does it mean if
 ?
? - What does it mean if
 ?
?
Review
Questions
- What becomes more than meaning every bit the pressure increases?
- Practise the bonny forces between gas particles get more prominent at higher or lower temperatures?
- Would HCl gas exist more than or less ideal than helium?
- real gas: A gas that does not bear according to the assumptions of the kinetic-molecular theory.
Dalton'southward Law of Fractional Pressures
- Define fractional pressure.
- State Dalton's law of partial pressures.
- Use this police to summate pressures of gas mixtures.

Is there oxygen available on Venus?
The atmosphere of Venus is markedly different from that of Earth. The gases in the Venusian atmosphere are 96.5% carbon dioxide and 3% nitrogen. The atmospheric pressure on Venus is roughly 92 times that of World, so the amount of nitrogen on Venus would contribute a force per unit area well over 2700 mm Hg. And there is no oxygen present, so we couldn't exhale there. Not that we would desire to become to Venus – the surface temperature is usually over 460°C.
Dalton'southward Constabulary of Fractional Pressures
Gas pressure level results from collisions betwixt gas particles and the inside walls of their container. If more gas is added to a rigid container, the gas pressure level increases. The identities of the ii gases do not matter. John Dalton, the English pharmacist who proposed the diminutive theory, too studied mixtures of gases. He found that each gas in a mixture exerts a pressure independently of every other gas in the mixture. For instance, our atmosphere is composed of about 78% nitrogen and 21% oxygen, with smaller amounts of several other gases making up the balance. Since nitrogen makes up 78% of the gas particles in a given sample of air, information technology exerts 78% of the pressure level. If the overall atmospheric pressure is one.00 atm, then the pressure of only the nitrogen in the air is 0.78 atm. The pressure of the oxygen in the air is 0.21 atm.
The partial pressure of a gas is the contribution that gas makes to the total pressure when the gas is part of a mixture. The fractional pressure of a gas is indicated by a ![]() with a subscript that is the symbol or formula of that gas. The partial pressure of nitrogen is represented by
with a subscript that is the symbol or formula of that gas. The partial pressure of nitrogen is represented by ![]() . Dalton's law of partial pressures states that the total pressure of a mixture of gases is equal to the sum of all of the partial pressures of the component gases. Dalton'south police tin be expressed with the post-obit equation:
. Dalton's law of partial pressures states that the total pressure of a mixture of gases is equal to the sum of all of the partial pressures of the component gases. Dalton'south police tin be expressed with the post-obit equation:
![]()
The Figure beneath shows two gases that are in split, equal-sized containers at the same temperature and pressure. Each exerts a different pressure, ![]() and
and ![]() , reflective of the number of particles in the container. On the right, the two gases are combined into the same container, with no volume change. The total pressure of the gas mixture is equal to the sum of the individual pressures. If
, reflective of the number of particles in the container. On the right, the two gases are combined into the same container, with no volume change. The total pressure of the gas mixture is equal to the sum of the individual pressures. If ![]() and
and ![]() , so
, so ![]() .
.

Figure xiv.13
Dalton's police says that the pressure of a gas mixture is equal to the partial pressures of the combining gases.
Summary
- The total pressure in a arrangement is equal to the sums of the fractional pressures of the gases present.
Do
Review the concepts at the link beneath and work the sample bug:
http://world wide web.kentchemistry.com/links/GasLaws/dalton.htm
Review
Questions
- What is the foundation for Dalton'south law?
- Argon makes upward about 0.93% of our atmosphere. If the atmospheric pressure is 760 mm Hg, what is the force per unit area contributed by argon?
- On a given twenty-four hour period, the h2o vapor in the air is 2.5%. If the partial pressure of the vapor is 19.4 mm Hg, what is the atmospheric pressure?
- Fractional pressure: The contribution that gas makes to the total pressure when the gas is part of a mixture.
- Dalton'south police of fractional pressures: The total pressure of a mixture of gases is equal to the sum of all of the fractional pressures of the component gases.
Mole Fraction
- Define mole fraction.
- Perform calculations involving mole fractions.

The mixed blessing of sulfur dioxide
Sulfur dioxide is a by-production of many processes, both natural and human-made. Massive amounts of this gas are released during volcanic eruptions such as the one seen above on the Large Island (Hawaii). Humans produce sulfur dioxide by called-for coal. The gas has a cooling effect when in the temper by reflecting sunlight dorsum away from the globe. Yet, sulfur dioxide is also a component of smog and acid rain, both of which are harmful to the environment. Many efforts have been made to reduce SO 2 levels to lower acid rain production. An unforeseen complication: as we lower the concentration of this gas in the atmosphere, we lower its ability to cool and then nosotros have global warming concerns.
Mole Fraction
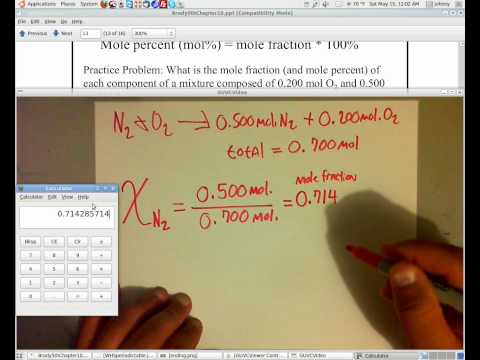
One style to express relative amounts of substances in a mixture is with the mole fraction. Mole fraction ![]() is the ratio of moles of ane substance in a mixture to the total number of moles of all substances. For a mixture of 2 substances,
is the ratio of moles of ane substance in a mixture to the total number of moles of all substances. For a mixture of 2 substances, ![]() and
and ![]() , the mole fractions of each would be written equally follows:
, the mole fractions of each would be written equally follows:
![]()
If a mixture consists of 0.fifty mol ![]() and 1.00 mol
and 1.00 mol ![]() , then the mole fraction of
, then the mole fraction of ![]() would be
would be ![]() . Similarly, the mole fraction of
. Similarly, the mole fraction of ![]() would exist
would exist ![]() .
.
Mole fraction is a useful quantity for analyzing gas mixtures in conjunction with Dalton's law of fractional pressures. Consider the post-obit situation: A 20.0 liter vessel contains 1.0 mol of hydrogen gas at a pressure of 600 mmHg. Some other twenty.0 liter vessel contains iii.0 mol of helium at a pressure of 1800 mmHg. These two gases are mixed together in an identical 20.0 liter vessel. Considering each volition exert its own pressure according to Dalton's law, we tin can limited the partial pressures as follows:
![]()
The partial pressure of a gas in a mixture is equal to its mole fraction multiplied past the total pressure. For our mixture of hydrogen and helium:
![]()
The total force per unit area according to Dalton'southward law is ![]() . So, each partial pressure level volition be:
. So, each partial pressure level volition be:
![]()
The partial pressures of each gas in the mixture don't alter since they were mixed into the aforementioned size vessel and the temperature was not changed.
Sample Problem: Dalton'southward Police force
A flask contains a mixture of 1.24 moles of hydrogen gas and 2.91 moles of oxygen gas. If the total pressure is 104 kPa, what is the partial pressure of each gas?
Step 1: List the known quantities and plan the problem .
Known
- 1.24 mol H 2
- 2.91 mol O two
-

Unknown
First, the mole fraction of each gas can exist determined. So, the partial pressure tin can be calculated past multiplying the mole fraction by the full force per unit area.
Pace 2: Solve .

Stride iii: Recollect most your result .
The hydrogen is slightly less than i 3rd of the mixture, so it exerts slightly less than 1 third of the total pressure.
Summary
- Apply of the mole fraction allows calculation to be made for mixtures of gases.
Practise
Questions
Watch the video at the link below and answer the following questions:
Click on the image higher up for more content
http://www.youtube.com/watch?v=7BaX__s-4Ls
- What is mole percent?
- Do the mole fractions add upwardly to 1.00?
- What other way could yous calculate the mole fraction of oxygen once you have the mole fraction of nitrogen?
Review
Questions
- What is mole fraction?
- How do you determine partial pressure level of a gas when given the mole fraction and the total pressure?
- In a gas mixture containing equal numbers of moles of ii gases, what can you lot say about the partial pressures of each gas?
- Mole fraction
 : The ratio of moles of one substance in a mixture to the total number of moles of all substances.
: The ratio of moles of one substance in a mixture to the total number of moles of all substances.
Gas Collection past Water Displacement
- Summate volumes of dry gases obtained after collecting over water.

What is the pressure?
Yous demand to do a lab experiment where hydrogen gas is generated. In social club to calculate the yield of gas, you have to know the pressure within the tube where the gas is collected. Simply how tin you go a barometer in there? Very simple: you don't. All y'all need is the atmospheric pressure in the room. As the gas pushed out the h2o, it is pushing against the atmosphere, so the pressure inside is equal to the pressure outside.
Gas Collection by Water Displacement
Gases that are produced in laboratory experiments are often collected by a technique called h2o displacement (see Figure beneath ). A canteen is filled with water and placed upside-down in a pan of water. The reaction flask is fitted with rubber tubing which is then fed under the bottle of h2o. As the gas is produced in the reaction flask, it exits through the rubber tubing and displaces the water in the bottle. When the bottle is full of the gas, it can be sealed with a lid.

Effigy xiv.xiv
A gas produced in a chemic reaction can be nerveless by water deportation.
Considering the gas is collected over water, it is not pure simply is mixed with vapor from the evaporation of the water. Dalton's law can be used to summate the amount of the desired gas by subtracting the contribution of the water vapor.
![]()
In society to solve a trouble, information technology is necessary to know the vapor pressure of water at the temperature of the reaction (see Table beneath). The sample trouble illustrates the utilise of Dalton'southward law when a gas is nerveless over h2o.
| Vapor Pressure of Water (mmHg) at Selected Temperatures (°C) | |||
|---|---|---|---|
| Temperature (°C) | Vapor Pressure (mmHg) | Temperature (°C) | Vapor Pressure (mmHg) |
| 0 | 4.58 | twoscore | 55.32 |
| 5 | 6.54 | 45 | 71.88 |
| x | 9.21 | 50 | 92.51 |
| fifteen | 12.79 | 55 | 118.04 |
| xx | 17.54 | 60 | 149.38 |
| 25 | 23.76 | 65 | 187.54 |
| 30 | 31.82 | 70 | 233.vii |
| 35 | 42.18 | ||
Sample Problem: Gas Collected past H2o Displacement
A certain experiment generates 2.58 L of hydrogen gas, which is collected over water. The temperature is 20°C and the atmospheric force per unit area is 98.60 kPa. Find the volume that the dry hydrogen would occupy at STP.
Footstep 1: List the known quantities and plan the trouble.
Known
Unknown
The atmospheric pressure is converted from kPa to mmHg in order to lucifer units with the table. The sum of the pressures of the hydrogen and the water vapor is equal to the atmospheric pressure. The pressure level of the hydrogen is institute by subtraction. And then, the volume of the gas at STP can be calculated by using the combined gas constabulary.
Step 2: Solve.
![]()
Now the combined gas law is used, solving for ![]() , the volume of hydrogen at STP.
, the volume of hydrogen at STP.
![]()
Step 3: Think near your result.
If the hydrogen gas were to exist collected at STP and without the presence of the water vapor, its volume would be 2.28 Fifty. This is less than the bodily collected book because some of that is h2o vapor. The conversion using STP is useful for stoichiometry purposes.
Summary
- The vapor pressure level due to water in a sample can be corrected for in lodge to get the true value for the pressure of the gas.
Practice
Questions
Watch the video at the link below and answer the post-obit questions:

Click on the image above for more content
http://world wide web.youtube.com/watch?5=xmL2Pax4yUQ
- What was the thistle tube used for?
- How did the instructor tests for oxygen?
- Did you notice whatever unsafe lab practices in the video?
- What would have happened to the splint if carbon dioxide had been nerveless?
Review
Questions
- Why is gas nerveless over water not pure?
- Why would we desire to correct for water vapor?
- A student wants to collect his gas over diethyl ether (vapor pressure of 530 mm Hg at 25°C). Is this a good idea? Explain your answer.
- water displacement: Collection of a gas over water.
Diffusion and Effusion and Graham's Constabulary
- Ascertain improvidence and effusion.
- State Graham's constabulary.
- Utilise Graham's constabulary to perform calculations involving movement of gases.

How exercise we know how fast a gas moves?
We usually cannot come across gases, so we demand means to observe their movements indirectly. The relative rates of diffusion of ammonia to hydrogen chloride tin be observed in a simple experiment. Cotton fiber balls are soaked with solutions of ammonia and hydrogen chloride (hydrochloric acrid) and attached to ii different prophylactic stoppers. These are simultaneously plugged into either finish of a long glass tube. The vapors of each travel down the tube at dissimilar rates. Where the vapors meet, they react to form ammonium chloride (NH 4 Cl), a white solid that appears in the glass tube as a ring.
Graham's Law
When a person opens a bottle of perfume in one corner of a big room, information technology doesn't take very long for the odor to spread throughout the entire room. Molecules of the perfume evaporate and the vapor spreads out to fill up the entire infinite. Diffusion is the tendency of molecules to move from an expanse of loftier concentration to an area of low concentration until the concentration is uniform. While gases lengthened rather rapidly, liquids lengthened much more slowly. Solids essentially do not diffuse.
Video of bromine diffusion: http://www.youtube.com/scout?v=R_xDe004oTQ

Click on the image in a higher place for more content
A related process to improvidence is the effusion. Effusion is the process of a confined gas escaping through a tiny pigsty in its container. Effusion can be observed past the fact that a helium-filled balloon will stop floating and sink to the flooring later a twenty-four hour period or and so. This is considering the helium gas effuses through tiny pores in the airship. Both diffusion and effusion are related to the speed at which various gas molecules move. Gases that have a lower molar mass effuse and lengthened at a faster rate than gases that have a college molar mass.
Scottish chemist Thomas Graham (1805-1869) studied the rates of effusion and diffusion of gases. Graham's police states that the rate of effusion or diffusion of a gas is inversely proportional to the square root of the molar mass of the gas. Graham'southward constabulary can be understood past comparing two gases ( ![]() and
and ![]() ) at the same temperature, meaning the gases take the aforementioned kinetic energy. The kinetic energy of a moving object is given by the equation
) at the same temperature, meaning the gases take the aforementioned kinetic energy. The kinetic energy of a moving object is given by the equation ![]() ,
,
where ![]() is mass and
is mass and ![]() is velocity. Setting the kinetic energies of the two gases equal to one another gives:
is velocity. Setting the kinetic energies of the two gases equal to one another gives:
![]()
The equation tin be rearranged to solve for the ratio of the velocity of gas ![]() to the velocity of gas
to the velocity of gas ![]() .
.
![]()
For the purposes of comparison the rates of effusion or improvidence of ii gases at the same temperature, the molar masses of each gas can exist used in the equation for ![]() .
.
Sample Problem: Graham's Law
Summate the ratio of diffusion rates of ammonia gas (NH three ) to hydrogen chloride (HCl) at the same temperature and pressure.
Step ane: Listing the known quantities and plan the problem.
Known
- molar mass NH 3 = 17.04 grand/mol
- tooth mass HCl = 36.46 g/mol
Unknown
- velcoity ratio

Substitute the molar masses of the gases into Graham'due south law and solve for the ratio.
Pace two: Solve.

The rate of diffusion of ammonia is 1.46 times faster than the charge per unit of diffusion of hydrogen chloride.
Pace 3: Think about your event
Since ammonia has a smaller molar mass than hydrogen chloride, the velocity of its molecules is greater and the velocity ratio is larger than ane.
Summary
- The processes of gas diffusion and effusion are described.
- Graham's law relates the molecular mass of a gas to its rate of improvidence or effusion.
Practice
Read the material on the link below and do the practice problems:
http://www.kentchemistry.com/links/GasLaws/GrahamsLaw.htm
Review
Questions
- Why tin can you scent nutrient cooking when you are in the side by side room?
- Why does a helium-filled balloon gradually sink?
- What does temperature accept to do with gas kinetic energies?
- improvidence: The tendency of molecules to move from an area of high concentration to an area of low concentration until the concentration is uniform.
- effusion: The process of a bars gas escaping through a tiny hole in its container.
- Graham'south law: The rate of effusion or diffusion of a gas is inversely proportional to the square root of the molar mass of the gas.
Source: https://courses.lumenlearning.com/cheminter/chapter/the-behavior-of-gases/
0 Response to "what will cause the size of the space in the top of the small tube to change in volume"
Postar um comentário