Can You Increase Chances of Gender of Baby by Timing Against Menstrual Cycle

The menstrual bike is a serial of natural changes in hormone production and the structures of the uterus and ovaries of the female reproductive arrangement that make pregnancy possible. The ovarian bicycle controls the production and release of eggs and the cyclic release of estrogen and progesterone. The uterine bicycle governs the preparation and maintenance of the lining of the uterus (womb) to receive a fertilized egg. These cycles are concurrent and coordinated, ordinarily last betwixt 21 and 35 days in developed women, with a median length of 28 days, and go on for near xxx–45 years.
Naturally occurring hormones drive the cycles; the cyclical rise and fall of the follicle stimulating hormone prompts the production and growth of oocytes (young egg cells). The hormone estrogen stimulates the uterus lining to thicken to adjust an embryo should fertilization occur. The claret supply of the thickened lining (endometrium) provides nutrients to a successfully implanted embryo. If implantation does not occur, the lining breaks down and blood is released. Triggered by falling progesterone levels, menses (a "period", in common parlance) is the cyclical shedding of the lining, and is a sign that pregnancy has not occurred.
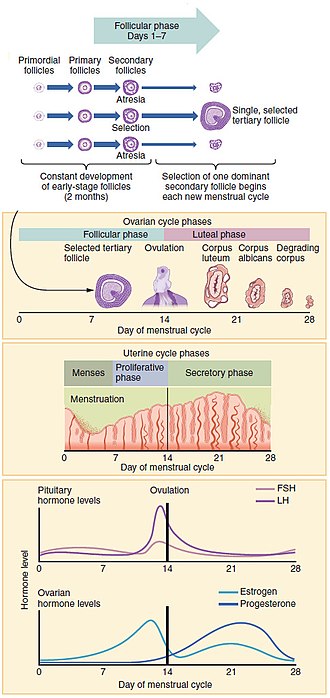
Each wheel occurs in phases based on events in the ovary (ovarian cycle) or the uterus (uterine bicycle). The ovarian wheel consists of the follicular stage, ovulation, and the luteal phase; the uterine cycle consists of the menstrual, proliferative and secretory phases. Day one of the menstrual cycle is the first day of the catamenia, which lasts for about five days. Around day fourteen, an egg is commonly released from the ovary. Menarche (the onset of the first menses) usually occurs around the age of twelve years.
The menstrual wheel can crusade some women to experience problems that disrupt daily lives. These tin can include cramps, tender breasts, tiredness, and premenstrual syndrome. More severe problems such as premenstrual dysphoric disorder are experienced by 3–8% of women. The menstrual cycle can be modified by hormonal nativity control.
Cycles and phases [edit]
Progression of the menstrual wheel and some of the hormones contributing to it
The menstrual cycle encompasses the ovarian and uterine cycles. The ovarian cycle describes changes that occur in the follicles of the ovary,[i] whereas the uterine wheel describes changes in the endometrial lining of the uterus. Both cycles tin be divided into phases. The ovarian cycle consists of alternating follicular and luteal phases, and the uterine cycle consists of menstruation, the proliferative phase, and the secretory phase.[ii] The menstrual cycle is controlled by the hypothalamus and the pituitary gland in the brain. The hypothalamus releases gonadotropin-releasing hormone (GnRH), which causes the nearby anterior pituitary gland to release follicle-stimulating hormone (FSH) and luteinizing hormone (LH). Before puberty, GnRH is released in low steady quantities and at a steady rate. Afterward puberty, GnRH is released in big pulses, and the frequency and magnitude of these determine how much FSH and LH are produced past the pituitary.[3]
Measured from the starting time day of one menstruation to the offset twenty-four hour period of the next, the length of a menstrual wheel varies only has a median length of 28 days.[four] The cycle is often less regular at the commencement and end of a woman'southward reproductive life.[4] At puberty, a child's trunk begins to mature into an adult body capable of sexual reproduction; the starting time menses (called menarche) occurs at around 12 years of age and continues for about 30–45 years.[5] [6] Menstrual cycles end at menopause, which is usually between 45 and 55 years of age.[7] [8]
Ovarian cycle [edit]
Between menarche and menopause the human ovaries regularly alternate between luteal and follicular phases during the monthly menstrual bike.[ix] Stimulated by gradually increasing amounts of estrogen in the follicular stage, discharges of blood period cease and the uterine lining thickens. Follicles in the ovary brainstorm developing nether the influence of a complex coaction of hormones, and after several days 1, or occasionally two, become dominant, while not-ascendant follicles shrink and die. Well-nigh mid-cycle, some 10–12 hours later the luteinizing hormone (LH) surges,[4] the dominant follicle releases an oocyte, in an event chosen ovulation.[10]
Afterwards ovulation, the oocyte lives for 24 hours or less without fertilization,[11] while the remains of the ascendant follicle in the ovary become a corpus luteum – a body with the master function of producing large amounts of the hormone progesterone.[12] [a] Under the influence of progesterone, the uterine lining changes to prepare for potential implantation of an embryo to establish a pregnancy. The thickness of the endometrium continues to increase in response to mounting levels of estrogen, which is released by the antral follicle (a mature ovarian follicle) into the blood apportionment. Elevation levels of estrogen are reached at effectually day thirteen of the bicycle and coincide with ovulation. If implantation does non occur within almost 2 weeks, the corpus luteum degenerates into the corpus albicans, which does not produce hormones, causing a precipitous drop in levels of both progesterone and estrogen. This drib causes the uterus to lose its lining in menstruation; information technology is around this fourth dimension that the lowest levels of estrogen are reached.[14]
In an ovulatory menstrual cycle, the ovarian and uterine cycles are concurrent and coordinated and concluding between 21 and 35 days in an adult woman, with a population boilerplate of 27–29 days.[fifteen] Although the boilerplate length of the man menstrual wheel is similar to that of the lunar cycle, in that location is no causal relation between the 2.[sixteen]
Follicular phase [edit]
The ovaries contain a finite number of egg stem cells, granulosa cells and theca cells, which together grade primordial follicles.[12] At effectually twenty weeks into gestation some 7 one thousand thousand immature eggs have already formed in an ovary. This decreases to around 2 one thousand thousand by the fourth dimension a girl is built-in, and 300,000 by the time she has her commencement period. On boilerplate, one egg matures and is released during ovulation each month later menarche.[17] Kickoff at puberty, these mature to primary follicles independently of the menstrual cycle.[18] The evolution of the egg is chosen oogenesis and only one jail cell survives the divisions to wait fertilization. The other cells are discarded as polar bodies, which cannot be fertilized.[xix] The follicular phase is the first office of the ovarian cycle and it ends with the completion of the antral follicles.[9] Meiosis (cell division) remains incomplete in the egg cells until the antral follicle is formed. During this stage normally only one ovarian follicle fully matures and gets set up to release an egg.[20] The follicular phase shortens significantly with age, lasting around 14 days in women aged eighteen–24 compared with 10 days in women aged forty–44.[14]
Through the influence of a ascension in follicle stimulating hormone (FSH) during the offset days of the cycle, a few ovarian follicles are stimulated. These follicles, which have been developing for the better part of a year in a process known as folliculogenesis, compete with each other for dominance. All but i of these follicles volition terminate growing, while ane dominant follicle – the one that has the nigh FSH receptors – volition continue to maturity. The remaining follicles die in a process called follicular atresia.[21] Luteinizing hormone (LH) stimulates further development of the ovarian follicle. The follicle that reaches maturity is called an antral follicle, and it contains the ovum (egg jail cell).[22]
The theca cells develop receptors that bind LH, and in response secrete big amounts of androstenedione. At the aforementioned time the granulosa cells surrounding the maturing follicle develop receptors that demark FSH, and in response start secreting androstenedione, which is converted to estrogen by the enzyme aromatase. The estrogen inhibits further production of FSH and LH by the pituitary gland. This negative feedback regulates levels of FSH and LH. The dominant follicle continues to secrete estrogen, and the rising estrogen levels make the pituitary more than responsive to GnRH from the hypothalamus. Equally estrogen increases this becomes a positive feedback signal, which makes the pituitary secrete more than FSH and LH. This surge of FSH and LH usually occurs one to two days before ovulation and is responsible for stimulating the rupture of the antral follicle and release of the oocyte.[18] [23]
Ovulation [edit]

An ovary about to release an egg
Effectually twenty-four hour period fourteen, the egg is released from the ovary.[24] Called "ovulation", this occurs when a mature egg is released from the ovarian follicles into the fallopian tube, almost 10–12 hours after the elevation in LH surge.[4] Typically only one of the 15–20 stimulated follicles reaches full maturity, and just i egg is released.[25] Ovulation just occurs in around 10% of cycles during the first ii years following menarche, and by the age of 40–50, the number of ovarian follicles is depleted.[26] LH initiates ovulation at around day 14 and stimulates the formation of the corpus luteum.[2] Post-obit further stimulation by LH, the corpus luteum produces and releases estrogen, progesterone, relaxin (which relaxes the uterus by inhibiting contractions of the myometrium), and inhibin (which inhibits further secretion of LH).[22]
The release of LH matures the egg and weakens the follicle wall in the ovary, causing the fully developed follicle to release its oocyte.[27] If it is fertilized past a sperm, the oocyte promptly matures into an ootid, which blocks the other sperm cells and becomes a mature egg. If it is not fertilized past a sperm, the oocyte degenerates. The mature egg has a bore of virtually 0.1 mm (0.0039 in),[28] and is the largest homo cell.[29]
Which of the two ovaries – left or right – ovulates appears random;[30] no left and correct coordinating process is known.[31] Occasionally both ovaries release an egg; if both eggs are fertilized, the consequence is fraternal twins.[32] Subsequently release from the ovary, the egg is swept into the fallopian tube by the fimbria – a fringe of tissue at the cease of each fallopian tube. After about a day, an unfertilized egg disintegrates or dissolves in the fallopian tube, and a fertilized egg reaches the uterus in 3 to five days.[33]
Fertilization unremarkably takes place in the ampulla, the widest department of the fallopian tubes. A fertilized egg immediately starts the procedure of embryogenesis (development). The developing embryo takes about iii days to reach the uterus, and some other iii days to implant into the endometrium. Information technology has usually reached the blastocyst stage at the fourth dimension of implantation: this is when pregnancy begins.[34] The loss of the corpus luteum is prevented by fertilization of the egg. The syncytiotrophoblast (the outer layer of the resulting embryo-containing blastocyst that later becomes the outer layer of the placenta) produces man chorionic gonadotropin (hCG), which is very similar to LH and preserves the corpus luteum. During the first few months of pregnancy, the corpus luteum continues to secrete progesterone and estrogens at slightly higher levels than those at ovulation. After this and for the rest of the pregnancy, the placenta secretes high levels of these hormones – along with human chorionic gonadotropin (hCG), which stimulates the corpus luteum to secrete more than progesterone and estrogens, blocking the menstrual cycle.[35] These hormones too set up the mammary glands for milk[b] production.[35]
Luteal phase [edit]
Lasting about 14 days,[4] the luteal phase is the concluding phase of the ovarian bicycle and information technology corresponds to the secretory phase of the uterine cycle. During the luteal stage, the pituitary hormones FSH and LH cause the remaining parts of the dominant follicle to transform into the corpus luteum, which produces progesterone.[37] [c] The increased progesterone starts to induce the production of estrogen. The hormones produced by the corpus luteum likewise suppress production of the FSH and LH that the corpus luteum needs to maintain itself. The level of FSH and LH fall quickly, and the corpus luteum atrophies.[39] Falling levels of progesterone trigger menstruation and the get-go of the side by side cycle. From the time of ovulation until progesterone withdrawal has acquired menstruation to begin, the process typically takes about two weeks. For an individual adult female, the follicular stage ofttimes varies in length from cycle to cycle; by contrast, the length of her luteal phase volition be fairly consistent from cycle to cycle at x to 16 days (average 14 days).[14]
Uterine cycle [edit]

The beefcake of the uterus
The uterine cycle has three phases: period, proliferative and secretory.[40]
Flow [edit]
Catamenia (besides called menstrual haemorrhage, menses or a flow) is the starting time and most evident phase of the uterine cycle and starting time occurs at puberty. Chosen menarche, the showtime catamenia occurs at the age of around twelve or 13 years.[8] The average age is generally afterwards in the developing globe and earlier in adult world.[41] In precocious puberty, it can occur as early on equally age 8 years,[42] and this can however be normal.[43] [44]
Menstruation is initiated each month past falling levels of estrogen and progesterone and the release of prostaglandins,[20] which constrict the spiral arteries. This causes them to spasm, contract and interruption up.[45] The blood supply to the endometrium is cutting off and the cells of the top layer of the endometrium (the stratum functionalis) become deprived of oxygen and dice. Later the whole layer is lost and only the bottom layer, the stratum basalis, is left in place.[20] An enzyme called plasmin breaks up the claret clots in the menstrual fluid, which eases the flow of blood and broken downwardly lining from the uterus.[46] The flow of blood continues for ii–6 days and effectually 30–lx milliliters of blood is lost,[fifteen] and is a sign that pregnancy has not occurred.[47]
The menstruum of claret normally serves every bit a sign that a woman has not become significant, but this cannot be taken as certainty, as several factors can cause bleeding during pregnancy.[48] Period occurs on boilerplate in one case a month from menarche to menopause, which corresponds with a woman'southward fertile years. The average age of menopause in women is 52 years, and it typically occurs between 45 and 55 years of age.[49] Menopause is preceded past a stage of hormonal changes called perimenopause.[7]
Eumenorrhea denotes normal, regular menses that lasts for around the start 5 days of the wheel.[24] Women who experience menorrhagia (heavy menstrual bleeding) are more susceptible to iron deficiency than the boilerplate person.[50]
Proliferative phase [edit]

During the menstrual cycle, levels of estradiol (an estrogen) vary by 200 pct. Levels of progesterone vary past over 1200 percentage.[51]
The proliferative phase is the second phase of the uterine cycle when estrogen causes the lining of the uterus to grow and proliferate.[39] The latter part of the follicular phase overlaps with the proliferative phase of the uterine cycle.[30] As they mature, the ovarian follicles secrete increasing amounts of estradiol, an estrogen. The estrogens initiate the formation of a new layer of endometrium in the uterus with the screw arterioles.[2]
As estrogen levels increase, cells in the cervix produce a type of cervical mucus[52] that has a higher pH and is less gluey than usual, rendering it more friendly to sperm.[53] This increases the chances of fertilization, which occurs around mean solar day xi to mean solar day 14.[eleven] This cervical fungus can be detected as a vaginal discharge that is copious and resembles raw egg whites.[54] For women who are practicing fertility awareness, it is a sign that ovulation may exist virtually to have place,[54] but it does not mean ovulation will definitely occur.[fifteen]
Secretory phase [edit]
The secretory phase is the last phase of the uterine cycle and information technology corresponds to the luteal phase of the ovarian cycle. During the secretory phase, the corpus luteum produces progesterone, which plays a vital role in making the endometrium receptive to the implantation of a blastocyst (a fertilized egg, which has begun to grow).[55] Glycogen, lipids, and proteins are secreted into the uterus[56] and the cervical mucus thickens.[57] In early pregnancy progesterone also increases blood period and reduces the contractility of the smooth muscle in the uterus[22] and raises the woman's basal torso temperature.[58]
If pregnancy does non occur the ovarian and uterine cycles outset over again.[46]
Anovulatory cycles and brusk luteal phases [edit]
Only ii thirds of overtly normal menstrual cycles are ovulatory, that is, cycles in which ovulation occurs.[fifteen] The other third lack ovulation or take a brusque luteal stage (less than 10 days[59]) in which progesterone production is bereft for normal physiology and fertility.[60] Cycles in which ovulation does not occur (anovulation) are common in girls who have just begun menstruating and in women around menopause. During the first ii years following menarche, ovulation is absent in effectually half of cycles. 5 years after menarche, ovulation occurs in around 75% of cycles and this reaches 80% in the following years.[61] Anovulatory cycles are often overtly identical to normally ovulatory cycles.[62] Any alteration to balance of hormones can lead to anovulation. Stress, anxiety and eating disorders can crusade a fall in GnRH, and a disruption of the menstrual cycle. Chronic anovulation occurs in six–15% of women during their reproductive years. Around menopause, hormone feedback dysregulation leads to anovulatory cycles. Although anovulation is not considered a disease, it can be a sign of an underlying condition such as polycystic ovary syndrome.[63] Anovulatory cycles or short luteal phases are normal when women are under stress or athletes increasing the intensity of training. These changes are reversible equally the stressors decrease or, in the case of the athlete, as she adapts to the preparation.[59]
Menstrual health [edit]

A human primary ovarian follicle viewed past microscopy. The round oocyte stained blood-red in the centre is surrounded by a layer of granulosa cells, which are enveloped by the basement membrane and theca cells. The magnification is around k times. (H&E stain)
Although a normal and natural procedure,[64] some women experience bug sufficient to disrupt their lives as a event of their menstrual cycle.[65] These include acne, tender breasts, feeling tired, and premenstrual syndrome (PMS).[65] [66] More severe bug such as premenstrual dysphoric disorder are experienced by 3 to 8% of women.[4] [67] Dysmenorrhea or "period pain"[68] tin cause cramps in the belly, back, or upper thighs that occur during the commencement few days of period.[69] Debilitating catamenia pain is not normal and tin can be a sign of something astringent such equally endometriosis.[70] These problems can significantly affect a adult female's health and quality of life and timely interventions tin improve the lives of these women.[71]
There are common culturally communicated misbeliefs that the menstrual cycle affects women's moods, causes depression or irritability, or that period is a painful, shameful or unclean experience. Often a woman'due south normal mood variation is falsely attributed to the menstrual cycle. Much of the research is weak, only there appears to exist a very small-scale increase in mood fluctuations during the luteal and menstrual phases, and a corresponding decrease during the rest of the cycle.[72] Changing levels of estrogen and progesterone across the menstrual wheel exert systemic effects on aspects of physiology including the brain, metabolism, and musculoskeletal system. The consequence tin can be subtle physiological and observable changes to women'southward athletic functioning including forcefulness, aerobic, and anaerobic performance.[73] Changes to the brain have also been observed throughout the menstrual wheel[74] but practise not interpret into measurable changes in intellectual achievement – including bookish functioning, problem-solving, memory, and creativity.[75] Improvements in spatial reasoning ability during the menstruation stage of the cycle are probably caused by decreases in levels of estrogen and progesterone.[72]
In some women, ovulation features a characteristic pain[d] called mittelschmerz (a High german term meaning middle pain). The cause of the hurting is associated with the ruptured follicle, causing a modest amount of claret loss.[20]
Even when normal, the changes in hormone levels during the menstrual bike can increase the incidence of disorders such as autoimmune diseases,[79] which might be caused by estrogen enhancement of the allowed organisation.[four]
Effectually xl% of women with epilepsy discover that their seizures occur more often at certain phases of their menstrual cycle. This catamenial epilepsy may be due to a drib in progesterone if it occurs during the luteal phase or around menstruation, or a surge in estrogen if it occurs at ovulation. Women who have regular periods tin can take medication just earlier and during menstruation. Options include progesterone supplements, increasing the dose of their regular anticonvulsant drug, or temporarily adding an anticonvulsant such equally clobazam or acetazolamide. If this is ineffective, or when a woman's menstrual cycle is irregular, then treatment is to stop the menstrual bicycle occurring. This may exist accomplished using medroxyprogesterone, triptorelin or goserelin, or past sustained use of oral contraceptives.[80] [81]
Hormonal contraception [edit]
Hormonal contraceptives forestall pregnancy by inhibiting the secretion of the hormones, FSH, LH and GnRH. Hormonal contraception that contains estrogen, such every bit combined oral contraceptive pills (COCs, oftentimes referred to every bit birth control pills) finish the development of the dominant follicle and the mid-cycle LH surge and thus ovulation.[82] Sequential dosing and discontinuation of the COC tin can mimic the uterine cycle and produce bleeding that resembles a period. In some cases, this bleeding is lighter.[83]
Progestin-only methods of hormonal contraception do not always preclude ovulation just instead work by stopping the cervical fungus from becoming sperm-friendly. Hormonal contraception is available in a variety of forms such as pills, patches, peel implants and hormonal intrauterine devices (IUDs).[84]
Evolution and other species [edit]
Almost female mammals accept an estrous bike, just simply 10 primate species, four bat species, the elephant shrews and the spiny mouse species Cairo spiny mouse (Acomys cahirinus) accept a menstrual wheel.[85] [86] The cycles are the aforementioned equally in humans apart from the length, which ranges from 9 to 37 days.[87] [85] The lack of immediate relationship betwixt these groups suggests that iv distinct evolutionary events take caused menstruation to arise.[88] In species that take a menstrual wheel, ovulation is not obvious to potential mates and at that place is no mating season.[89] [90] There are four theories on the evolutionary significance of catamenia:[88]
- Control of sperm-borne pathogens.[91] [92] [93] This hypothesis held that menstruation protected the uterus against pathogens introduced by sperm. Hypothesis i does not accept into business relationship that copulation tin can take place weeks before menstruation and that potentially infectious semen is not controlled by menstruum in other species.[88]
- Free energy conservation.[92] [94] This hypothesis claimed that it took less free energy to rebuild a uterine lining than to maintain it if pregnancy did non occur. Hypothesis 2 does non explain other species that likewise do not maintain a uterine lining but practice not menstruate.[88]
- A theory based on spontaneous decidualization (a procedure that results in significant changes to cells of the endometrium in training for, and during, pregnancy, in which the endometrium changes into the decidua). Decidualization leads to the development of the endothelium, which involves cells of the allowed system,[87] the formation of a new claret supply, hormones and tissue differentiation. In non-menstruating mammals, decidualization is driven by the embryo, non the mother.[92] It evolved in some placental mammals because it confers advantages in that it allows females to prepare for pregnancy without needing a signal from the fetus.[88] Hypothesis 3 defers to an explanation of the evolutionary origin of spontaneous decidualization and does not explicate the evolution of menstruation alone.[88]
- Uterine pre-conditioning.[95] This hypothesis claims that a monthly pre-workout of the uterus is needed in species, such every bit humans, that have securely invasive (deep-rooted) placentas. In the process leading to the formation of a placenta, maternal tissues are invaded. This hypothesis holds that menstruation was non evolutionary, rather the result of a casual pre-conditioning of the uterus to protect uterine tissue from the securely rooting placenta, in which a thicker endometrium develops.[95] Hypothesis 4 does not explain menses in not-primates.[88]
Notes [edit]
- ^ Progesterone levels exceed those of estrogen (estradiol) by a hundred-fold.[13]
- ^ Breastfeeding women can feel consummate suppression of follicular development, follicular development but no ovulation, or resumption of normal menstrual cycles.[36]
- ^ In the corpus luteum, cholesterol side-chain cleavage enzyme converts cholesterol to pregnenolone, which is converted to progesterone.[38]
- ^ Uncharacteristic mid-cycle pain may be caused by medical conditions such every bit ectopic pregnancy or ruptured ovarian cyst[76] [77] or may exist dislocated with appendicitis.[78]
References [edit]
- ^ Richards JS (2018). "The ovarian cycle". Vitamins and Hormones (Review). 107: one–25. doi:10.1016/bs.vh.2018.01.009. ISBN978-0-128-14359-nine. PMID 29544627.
- ^ a b c Tortora 2017, p. 944.
- ^ Prior 2020, p. 42.
- ^ a b c d e f g Reed BF, Carr BR, Feingold KR, et al. (2018). "The Normal Menstrual Cycle and the Control of Ovulation". Endotext (Review). PMID 25905282. Archived from the original on 28 May 2021. Retrieved 8 January 2021.
- ^ Prior 2020, p. 40.
- ^ Lacroix AE, Gondal H, Langaker MD (2020). "Physiology, menarche". StatPearls [Internet]. Treasure Isle (FL): StatPearls Publishing (Review). PMID 29261991.
- ^ a b Rodriguez-Landa 2017, p. 8.
- ^ a b Papadimitriou A (December 2016). "The evolution of the historic period at menarche from prehistorical to modernistic times". Periodical of Pediatric and Boyish Gynecology (Review). 29 (vi): 527–xxx. doi:10.1016/j.jpag.2015.12.002. PMID 26703478.
- ^ a b Sherwood 2016, p. 741.
- ^ Sherwood 2016, p. 747.
- ^ a b Tortora 2017, p. 957.
- ^ a b Tortora 2017, p. 929.
- ^ Prior 2020, p. 41.
- ^ a b c Tortora 2017, pp. 942–46.
- ^ a b c d Prior 2020, p. 45.
- ^ Norris & Carr 2013, p. 361.
- ^ Ugwumadu 2014, p. 115.
- ^ a b Watchman 2020, p. 8.
- ^ Schmerler S, Wessel GM (January 2011). "Polar bodies – more a lack of understanding than a lack of respect". Molecular Reproduction and Development (Review). 78 (1): 3–8. doi:10.1002/mrd.21266. PMC3164815. PMID 21268179.
- ^ a b c d Tortora 2017, p. 945.
- ^ Johnson 2007, p. 86.
- ^ a b c Tortora 2017, p. 942.
- ^ Sherwood 2016, p. 745.
- ^ a b Tortora 2017, p. 943.
- ^ Sadler 2019, p. 48.
- ^ Tortora 2017, p. 953.
- ^ Sherwood 2016, p. 746.
- ^ Alberts B, Johnson A, Lewis J, Raff M, Roberts G, Walter P (2002). "Eggs". Molecular Biology of the Prison cell (quaternary ed.). New York: Garland Science. ISBN0-8153-3218-i. Archived from the original on 16 December 2019. Retrieved 25 Feb 2021.
- ^ Iussig B, Maggiulli R, Fabozzi K, Bertelle S, Vaiarelli A, Cimadomo D, Ubaldi FM, Rienzi Fifty (May 2019). "A brief history of oocyte cryopreservation: Arguments and facts". Acta Obstetricia et Gynecologica Scandinavica (Review). 98 (five): 550–58. doi:x.1111/aogs.13569. PMID 30739329.
- ^ a b Parker 2019, p. 283.
- ^ Johnson 2007, pp. 192–93.
- ^ Johnson 2007, p. 192.
- ^ Sadler 2019, p. 36.
- ^ Tortora 2017, p. 959.
- ^ a b Tortora 2017, p. 976.
- ^ Carr SL, Gaffield ME, Dragoman MV, Phillips Due south (September 2016). "Safety of the progesterone-releasing vaginal band (PVR) among lactating women: A systematic review". Contraception (Review). 94 (3): 253–61. doi:ten.1016/j.contraception.2015.04.001. PMID 25869631.
- ^ Johnson 2007, p. 91.
- ^ Male monarch SR, LaVoie HA (January 2012). "Gonadal transactivation of STARD1, CYP11A1 and HSD3B". Frontiers in Bioscience (Landmark Edition). 17: 824–46. doi:x.2741/3959. PMID 22201776.
- ^ a b Ugwumadu 2014, p. 117.
- ^ Salamonsen LA (Dec 2019). "Women in reproductive scientific discipline: Agreement homo endometrial function". Reproduction (Cambridge, England) (Review). 158 (vi): F55–F67. doi:10.1530/REP-18-0518. PMID 30521482.
- ^ Alvergne A, Högqvist Tabor V (June 2018). "Is female wellness cyclical? Evolutionary perspectives on menstruation". Trends in Ecology & Evolution (Review). 33 (6): 399–414. arXiv:1704.08590. doi:x.1016/j.tree.2018.03.006. PMID 29778270. S2CID 4581833.
- ^ Ibitoye Yard, Choi C, Tai H, Lee G, Sommer Thou (2017). "Early on menarche: A systematic review of its issue on sexual and reproductive health in low- and middle-income countries". PLOS ONE (Review). 12 (6): e0178884. Bibcode:2017PLoSO..1278884I. doi:ten.1371/journal.pone.0178884. PMC5462398. PMID 28591132.
- ^ "Menstruation and the menstrual bike fact sheet". Office of Women'south Wellness. US Department of Wellness and Man Services. 23 December 2014. Archived from the original on 26 June 2015. Retrieved 25 June 2015.
- ^ Sultan C, Gaspari Fifty, Maimoun Fifty, Kalfa N, Paris F (April 2018). "Disorders of puberty" (PDF). Best Practice & Enquiry. Clinical Obstetrics & Gynaecology (Review). 48: 62–89. doi:10.1016/j.bpobgyn.2017.xi.004. PMID 29422239. Archived (PDF) from the original on 1 July 2020. Retrieved 27 February 2021.
- ^ Johnson 2007, p. 152.
- ^ a b Tortora 2017, p. 600.
- ^ Johnson 2007, p. 99.
- ^ Breeze C (May 2016). "Early on pregnancy bleeding". Australian Family unit Medico (Review). 45 (five): 283–86. PMID 27166462.
- ^ Towner MC, Nenko I, Walton SE (April 2016). "Why do women terminate reproducing before menopause? A life-history approach to age at final birth". Philosophical Transactions of the Royal Gild of London. Series B, Biological Sciences (Review). 371 (1692): 20150147. doi:10.1098/rstb.2015.0147. PMC4822427. PMID 27022074.
- ^ Harvey LJ, Armah CN, Prissy JR, Foxall RJ, John Lewis D, Langford NJ, Fairweather-Tait SJ (Oct 2005). "Affect of menstrual claret loss and diet on iron deficiency among women in the UK". The British Journal of Nutrition (Comparative study). 94 (4): 557–64. doi:10.1079/BJN20051493. PMID 16197581.
- ^ Prior JC (2020). "Women's reproductive organization as balanced estradiol and progesterone actions—A revolutionary, paradigm-shifting concept in women'south health". Drug Discovery Today: Disease Models. 32, Office B: 31–40. doi:10.1016/j.ddmod.2020.11.005.
- ^ Simmons RG, Jennings Five (July 2020). "Fertility awareness-based methods of family unit planning". All-time Practice & Enquiry. Clinical Obstetrics & Gynaecology (Review). 66: 68–82. doi:x.1016/j.bpobgyn.2019.12.003. PMID 32169418.
- ^ Tortora 2017, pp. 936–37.
- ^ a b Su HW, Yi YC, Wei TY, Chang TC, Cheng CM (September 2017). "Detection of ovulation, a review of currently available methods". Bioeng Transl Med (Review). 2 (3): 238–46. doi:10.1002/btm2.10058. PMC5689497. PMID 29313033.
- ^ Lessey BA, Young SL (Apr 2019). "What exactly is endometrial receptivity?". Fertility and Sterility (Review). 111 (4): 611–17. doi:10.1016/j.fertnstert.2019.02.009. PMID 30929718.
- ^ Salamonsen LA, Evans J, Nguyen HP, Edgell TA (March 2016). "The microenvironment of man implantation: determinant of reproductive success". American Periodical of Reproductive Immunology (Review). 75 (3): 218–25. doi:x.1111/aji.12450. PMID 26661899.
- ^ Han L, Taub R, Jensen JT (November 2017). "Cervical mucus and contraception: what we know and what we don't". Contraception (Review). 96 (5): 310–321. doi:10.1016/j.contraception.2017.07.168. PMID 28801053.
- ^ Charkoudian N, Hart EC, Barnes JN, Joyner MJ (June 2017). "Autonomic control of body temperature and blood pressure: influences of female sex hormones" (PDF). Clinical Autonomic Inquiry (Review). 27 (3): 149–55. doi:ten.1007/s10286-017-0420-z. hdl:1983/c0c1058c-553b-4563-8dd1-b047d9b672c1. PMID 28488202. S2CID 3773043. Archived (PDF) from the original on 10 May 2020. Retrieved 27 Feb 2021.
- ^ a b Liu AY, Petit MA, Prior JC (2020). "Exercise and the hypothalamus: ovulatory adaptations". In Hackney Air-conditioning, Constantini NW (eds.). Endocrinology of Physical Activeness and Sport. Contemporary Endocrinology. Springer International Publishing. pp. 124–47. doi:10.1007/978-3-030-33376-8_8. ISBN978-3-030-33376-eight. S2CID 243129220.
- ^ Prior 2020, p. 46.
- ^ Elmaoğulları S, Aycan Z (July 2018). "Abnormal uterine haemorrhage in adolescents". Journal of Clinical Research in Pediatric Endocrinology. 10 (iii): 191–97. doi:10.4274/jcrpe.0014. PMC6083466. PMID 29537383.
- ^ Prior 2020, p. 44.
- ^ Hernandez-Rey, AE (2 August 2018). "Anovulation". Medscape. Medscape LLC. Archived from the original on twenty March 2021. Retrieved 30 March 2021.
- ^ Prior 2020, p. fifty.
- ^ a b Gudipally PR, Sharma GK (2020). "Premenstrual syndrome". StatPearls [Internet] (Review). PMID 32809533.
- ^ Ferries-Rowe Eastward, Corey East, Archer JS (November 2020). "Principal Dysmenorrhea: Diagnosis and Therapy". Obstetrics and Gynecology. 136 (5): 1047–1058. doi:x.1097/AOG.0000000000004096. PMID 33030880.
- ^ Appleton SM (March 2018). "Premenstrual syndrome: show-based evaluation and treatment". Clinical Obstetrics and Gynecology (Review). 61 (one): 52–61. doi:x.1097/GRF.0000000000000339. PMID 29298169. S2CID 28184066.
- ^ Nagy H, Khan MA (2020). "Dysmenorrhea". StatPearls (Review). PMID 32809669.
- ^ Baker FC, Lee KA (September 2018). "Menstrual cycle effects on slumber". Sleep Medicine Clinics (Review). xiii (3): 283–94. doi:x.1016/j.jsmc.2018.04.002. PMID 30098748. S2CID 51968811.
- ^ Maddern J, Grundy 50, Castro J, Brierley SM (2020). "Hurting in endometriosis". Frontiers in Cellular Neuroscience. xiv: 590823. doi:10.3389/fncel.2020.590823. PMC7573391. PMID 33132854.
- ^ Matteson KA, Zaluski KM (September 2019). "Menstrual health equally a part of preventive wellness intendance". Obstetrics and Gynecology Clinics of North America (Review). 46 (3): 441–53. doi:10.1016/j.ogc.2019.04.004. PMID 31378287. S2CID 199437314.
- ^ a b Else-Quest & Hyde 2021, pp. 258–61.
- ^ Carmichael MA, Thomson RL, Moran LJ, Wycherley TP (February 2021). "The impact of menstrual cycle phase on athletes' performance: a narrative review". Int J Environ Res Public Health (Review). xviii (four): 1667. doi:10.3390/ijerph18041667. PMC7916245. PMID 33572406.
- ^ Pletzer B, Harris TA, Scheuringer A, Hidalgo-Lopez E (October 2019). "The cycling encephalon: menstrual cycle related fluctuations in hippocampal and fronto-striatal activation and connectivity during cognitive tasks". Neuropsychopharmacology. 44 (eleven): 1867–75. doi:10.1038/s41386-019-0435-iii. PMC6785086. PMID 31195407.
- ^ Le J, Thomas N, Gurvich C (March 2020). "Cognition, the menstrual wheel, and premenstrual disorders: a review". Brain Sci (Review). ten (four): 198. doi:x.3390/brainsci10040198. PMC7226433. PMID 32230889.
- ^ Kruszka PS, Kruszka SJ (July 2010). "Evaluation of acute pelvic hurting in women". Am Fam Medico (Review). 82 (ii): 141–47. PMID 20642266. Archived from the original on 27 Jan 2021. Retrieved 4 March 2021.
- ^ Cleary Yard, Flanagan KW (2019). Acute and Emergency Intendance in Able-bodied Preparation. Human Kinetics. p. 340.
- ^ Brott NR, Le JK (2020). "Mittelschmerz". Stat Pearls [Net] (Review). PMID 31747229. Archived from the original on 28 May 2021. Retrieved 4 March 2021.
- ^ Talsania K, Scofield RH (May 2017). "Menopause and rheumatic illness". Rheumatic Affliction Clinics of North America (Review). 43 (2): 287–302. doi:10.1016/j.rdc.2016.12.011. PMC5385852. PMID 28390570.
- ^ Maguire MJ, Nevitt SJ (September 2021). "Treatments for seizures in catamenial (menstrual-related) epilepsy". The Cochrane Database of Systematic Reviews. 2021 (9): CD013225. doi:10.1002/14651858.CD013225.pub3. PMC 8444032. PMID 34528245.
- ^ Sveinsson O, Tomson T (September 2014). "Epilepsy and menopause: potential implications for pharmacotherapy". Drugs & Aging. 31 (9): 671–75. doi:x.1007/s40266-014-0201-5. PMID 25079452. S2CID 21166687.
- ^ Tortora 2017, p. 948.
- ^ Polis CB, Hussain R, Berry A (June 2018). "There might be blood: a scoping review on women'southward responses to contraceptive-induced menstrual haemorrhage changes". Reproductive Health. 15 (1): 114. doi:10.1186/s12978-018-0561-0. PMC6020216. PMID 29940996.
- ^ Tortora 2017, pp. 948–49.
- ^ a b Bellofiore N, Ellery SJ, Mamrot J, Walker DW, Temple-Smith P, Dickinson H (January 2017). "Get-go evidence of a menstruating rodent: the spiny mouse (Acomys cahirinus)" (PDF). American Periodical of Obstetrics and Gynecology (Journal article). 216 (1): xl.e1–40.e11. doi:10.1016/j.ajog.2016.07.041. PMID 27503621. S2CID 88779.
- ^ Bellofiore, Nadia; Cousins, Fiona; Temple-Smith, Peter; Evans, Jemma (1 February 2019). "Altered exploratory behaviour and increased food intake in the spiny mouse before catamenia: a unique pre-clinical model for examining premenstrual syndrome". Human Reproduction. 34 (2): 308–322. doi:10.1093/humrep/dey360. ISSN 0268-1161. PMID 30561655.
- ^ a b Catalini Fifty, Fedder J (May 2020). "Characteristics of the endometrium in menstruating species: lessons learned from the fauna kingdom†". Biological science of Reproduction (Journal commodity). 102 (6): 1160–69. doi:ten.1093/biolre/ioaa029. PMC7253787. PMID 32129461.
- ^ a b c d e f g Emera D, Romero R, Wagner K (January 2012). "The evolution of menstruation: a new model for genetic absorption: explaining molecular origins of maternal responses to fetal invasiveness". BioEssays (Journal article). 34 (1): 26–35. doi:10.1002/bies.201100099. PMC3528014. PMID 22057551. See BBC Earth lay summary, xx Apr 2015.
- ^ Schjenken JE, Robertson SA (July 2020). "The female response to seminal fluid". Physiological Reviews (Review). 100 (3): 1077–117. doi:ten.1152/physrev.00013.2018. PMID 31999507. S2CID 210983017.
- ^ Muller MN (May 2017). "Testosterone and reproductive effort in male primates". Hormones and Behavior (Review). 91: 36–51. doi:ten.1016/j.yhbeh.2016.09.001. PMC5342957. PMID 27616559.
- ^ Martin RD (2007). "The evolution of man reproduction: a primatological perspective". American Journal of Physical Anthropology (Review). 134 (S45): 59–84. doi:10.1002/ajpa.20734. PMID 18046752.
- ^ a b c Finn CA (June 1998). "Catamenia: a nonadaptive effect of uterine development". The Quarterly Review of Biology (Review). 73 (two): 163–73. doi:ten.1086/420183. PMID 9618925. S2CID 25135630.
- ^ Profet M (September 1993). "Catamenia equally a defense against pathogens transported by sperm". The Quarterly Review of Biology (Review). 68 (3): 335–86. doi:10.1086/418170. PMID 8210311. S2CID 23738569.
- ^ Strassmann BI (June 1996). "The evolution of endometrial cycles and menstruation". The Quarterly Review of Biology (Review). 71 (2): 181–220. doi:10.1086/419369. PMID 8693059. S2CID 6207295.
- ^ a b Brosens JJ, Parker MG, McIndoe A, Pijnenborg R, Brosens IA (June 2009). "A role for menstruum in preconditioning the uterus for successful pregnancy". American Periodical of Obstetrics and Gynecology (Journal article). 200 (6): 615.e1–6. doi:10.1016/j.ajog.2008.11.037. PMID 19136085.
Book sources [edit]
- Else-Quest Due north, Hyde JS (2021). "Psychology, gender, and health: psychological aspects of the menstrual cycle". The Psychology of Women and Gender: Half the Human Experience + (10th ed.). Los Angeles: SAGE Publishing. ISBN978-i-544-39360-5.
- Johnson MH (2007). Essential Reproduction. Malden, Massachusetts: Wiley-Blackwell. ISBN978-1-4051-1866-8. OCLC 76074156.
- Norris DA, Carr JA (2013). Vertebrate Endocrinology (5th ed.). Academic Press. ISBN978-0-123-96465-half-dozen.
- Parker S (2019). The Curtailed Man Body Volume: An Illustrated Guide to its Structures, Function and Disorders. London: DK. ISBN978-0-241-39552-3. OCLC 1091644711.
- Prior JC (2020). "The menstrual cycle: its biology in the context of silent ovulatory disturbances". In Ussher JM, Chrisler JC, Perz J (eds.). Routledge International Handbook of Women'due south Sexual and Reproductive Health (1st ed.). Abingdon, Oxon: Routledge. ISBN978-1-138-49026-0. OCLC 1121130010.
- Rodriguez-Landa J (2017). A Multidisciplinary Look at Menopause. Rijeka, Croatia: IntechOpen. ISBN978-953-51-3405-3. OCLC 1193045564.
- Sadler TW (2019). Langman'southward Medical Embryology. Philadelphia: Wolters Kluwer. ISBN978-1-4963-8390-vii. OCLC 1042400100.
- Sherwood L (2016). Man Physiology: From Cells to Systems. Boston, Massachusetts: Cengage Learning. ISBN978-1-285-86693-2. OCLC 905848832.
- Tortora Thou (2017). Tortora's Principles of Anatomy & Physiology. Hoboken, New Jersey: John Wiley & Sons, Inc. ISBN978-1-119-38292-8. OCLC 990424568.
- Ugwumadu A (2014). Basic Sciences for Obstetrics and Gynaecology: Cadre Material for MRCOG. Oxford, England: Oxford University Printing. ISBN978-0-19-953508-8. OCLC 889303297.
- Watchman T (2020). Zero to Finals : Obstetrics and Gynaecology. Manchester: Zero to Finals. ISBN979-eight-6037-9726-seven. OCLC 1233034578.
External links [edit]
![]() Media related to Menstrual cycle at Wikimedia Commons
Media related to Menstrual cycle at Wikimedia Commons
Source: https://en.wikipedia.org/wiki/Menstrual_cycle
0 Response to "Can You Increase Chances of Gender of Baby by Timing Against Menstrual Cycle"
Postar um comentário